Abstract
Introduction: Hodgkin lymphoma (HL) is the most frequent malignancy in young adults. The high probability of the cure is redeemed by both acute and long-term treatment sequelae. Balancing toxicity and efficacy requires highly reliable decision-making tools. Current treatment tailoring is based on the positron-emission tomography (PET/CT) assessment. The way how to improve the sensitivity of imaging methods could be a detection of minimal residual disease (MRD) in the circulating-tumor DNA (ctDNA) samples. The standard of the detection method is a next-generation sequencing (NGS), which covers a large spectrum of mutations, but is time-consuming, costly, and requires a significant concentration of ctDNA. The new digital PCR (dPCR) is a faster, cheaper method, which needs a lower amount of ctDNA.
Aim: To validate the reliability of the dPCR for target-mutations of STAT6 and XPO1 genes detection in the peripheral blood ctDNA of patients with Hodgkin lymphoma
Methods: CtDNA samples were obtained at the time of diagnosis/relapse, at the time of PET/CT assessment (interim, final), and during follow-up - every 3-6 months.
CfDNA was extracted from peripheral blood plasma using the QiaAmp Circulating nucleic acid kit (Qiagen). Specific NGS panel covering coding sequences (including UTRs) of 13 selected genes (B2M, CD36, CIITA, GNA13, HIST1H1E, ITPKB, NFKBIE, PTPN1, SOCS1, SPEN, STAT6, TNFAIP3, and XPO1) was designed. For library preparation, we used SureSelect XT HS2 technology (Agilent Technologies) based on "target enrichment" with molecular barcodes. Sequencing was performed on a NovaSeq6000 (Illumina). Data were analyzed with the SureCall software (Agilent Technologies) with the sensitivity of 1.0% VAF. The detected variants were annotated using COSMIC, dbSNP, Ensembl, and ClinVar. Selected variants STAT6 p.N417Y and XPO1 p.E571K were further monitored by dPCR (QIAcuity Digital PCR System; Qiagen) with the sensitivity of 0.1% VAF. For dPCR experiments, we used a QIAcuityTM probe PCR kit (Qiagen) and nanoplate QIAcuity Nanoplate 26K 24-well (Qiagen). Primers and probes were taken and modified according to Camus et al., 2016 (EXPO1 p.E571K) and Spina et al., 2018 (STAT6 p.N417Y). All pts were analyzed with the same Siemens Biograph mCT scanner and results were independently reviewed by two physicians blinded for clinical outcome.
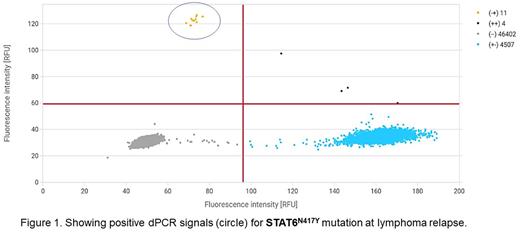
Results: We have analyzed ctDNA using an NGS panel in 48 pts with CHL (44 at diagnosis, 4 in relapse). Mutations were detected in 22/48 (46%) pts. The most frequently mutated genes were STAT6 (12/48 pts), TNFAIP3 (10/48 pts), XPO1 (7/48 pts), and SOCS1 (7/26 pts). Frameshift deletions prevailed in TNFAIP3 and SOCS1 genes. Hotspot mutations in the STAT6 (p.N417Y, in five pts) and XPO1 (p.E571K, in six pts) genes we used as target mutations for dPCR cohort. The first date of ctDNA analysis was the first CHL diagnosis in ten and 1st relapse in one case. This dPCR-cohort (n=11) consists of 9 females and 2 males with a median age at the time of diagnosis of 40 yrs, with disease subtypes of nodular sclerosis (8), mixed-cellularity (1), lymphocyte-rich (1), and CHL unclassified (1). Mean VAF at diagnosis was 9.14% (0.76-25.0), mean cell-free DNA concentration 1.47 ng/mL (0.76-2.88). Three of the pts were considered to have intermediate and eight advanced cHL stages according to the GHSG system. Pts have received ABVD (n=5) or BEACOPPesc (n=5) or platinum-based+BEAM200 (n=1) protocols. Interim PET-2 was positive in 1 pt (stable disease), and final PET was positive in 2 (both PD). dPCR captured mutation in all three PETpos (0.12, 0.86, and 2.22%) cases, whereas was negative (<0.1% VAF) in all PETneg cases. During follow-up, two patients relapsed (both with detectable mutation, illustrative case is a Figure 1), in one case (pt during the BV post-ASCT maintenance) we observed transient dPCR positivity without evidence of relapse.
Conclusion: A combination of the NGS (large-scale mutation screening) at the diagnosis with rapid and sensitive mutation monitoring using dPCR at the time of the treatment-tailoring or follow-up could improve decision-making accuracy in the routine care of Hodgkin lymphoma patients. Further analyses are needed to directly compare the sensitivity of the NGS and dPCR methods.
Acknowledgement: Supported by MZCR-RVO (FNOL, 00098892) and AZV NU22-03-0018 grants.
Disclosures
Prochazka:Hoffmann-La Roche: Speakers Bureau; Takeda: Speakers Bureau; Novartis: Speakers Bureau. Papajík:AstraZeneca: Honoraria, Research Funding; Abbvie: Honoraria, Research Funding; Novartis: Honoraria, Research Funding. Bachanova:FATA Therapeutics: Research Funding; Incyte: Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees; Citius Pharma: Research Funding; Karyopharma: Consultancy; Gamida Cell: Membership on an entity's Board of Directors or advisory committees, Research Funding; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal